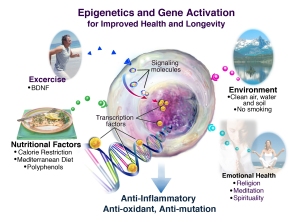
Recognizing the importance of diet is one of the incredible gifts that epigenetics has given us.
Recognizing the importance of diet is one of the incredible gifts that epigenetics has given us.
Incredible, because researchers have discovered, through pure science, that the best way to turn ON the “helpful” genes and turn OFF the “harmful” genes is through a proper diet.
There has never been an intersection point in modern history where scientists and nutritionists have met with such like minds.
Sure, most scientists are researching with the goal of developing a drug that will throw ON the good switches … but until those drugs are developed, the best methods they have discovered to turn on the good genes is a healthy and balanced diet, with the addition of some key supplements.
Many of their recommendations are listed in the excerpts below.
from The University of Utah Genetic Science Learning Center “Nutrition and the Epigenome.”
“Unlike behavior or stress, diet is one of the more easily studied, and therefore better understood, environmental factors in epigenetic change.
The nutrients we extract from food enter metabolic pathways where they are manipulated, modified and molded into molecules the body can use. One such pathway is responsible for making methyl groups – important epigenetic tags that silence genes.
Familiar nutrients like folic acid, B vitamins and SAM-e (S-Adenosyl methionine, a popular over-the-counter supplement) are key components of this methyl-making pathway. Diets high in these methyl-donating nutrients can rapidly alter gene expression, especially during early development when the epigenome is first being established.
 Diet During Early Development Can Cause Changes Lasting Into Adulthood
Diet During Early Development Can Cause Changes Lasting Into Adulthood
Your mother’s diet during pregnancy and what you’re fed as an infant can cause critical changes that stick with you into adulthood. Animal studies have shown that deficiency of methyl-donating folate or choline during late fetal or early postnatal development causes certain regions of the genome to be under-methylated for life.
For adults, a methyl deficient diet still leads to a decrease in DNA methylation, but the changes are reversible with resumption of a normal diet.
The Emerging Field of Nutrigenomics
As we better understand the connections between diet and the epigenome, the opportunity arises for clinical applications. Just as mapping our gene variations gives us a window into our personalized medical needs, so might a profile of one’s unique epigenome.
Formed through a lifetime of experiences beginning in the womb, our epigenome may provide a wealth of information about how to eat better. Enter the future field of nutrigenomics, where nutritionists take a look at your methylation pattern and design a personalized nutrition plan. While we’re not quite to that point yet, your doctor can already tell a lot about the your disease risk by looking at your family health history.”
from “Epigenetics, Methylation, and Gene Expression.” by Kevin Cann, April 10th, 2013
“The elasticity of our epigenome is critical for human survival. It allows us to adapt and survive in changing environments. Diet and stress play an important role in gene expression. An interesting study just came out of UMASS Medical School and was published in the journal Cell regarding diet and gene expression. The researchers concluded their study by saying even the smallest amount of food can alter gene expression. The interesting piece is that the gene regulators that were affected by nutrition in the study also play a role in maintaining human circadian rhythm, and they react very quickly to food choices. This is important to the individuals that are genetically predisposed to obesity or disease and/or making lifestyle choices that are causing negative gene expression. Everything in moderation is not the key. This is also important for those that may have a genetic disorder.
Altered circadian rhythm and increased oxidative stress are largely responsible for the onset of symptoms seen in Huntington’s Disease. This concept becomes extremely important for those that tested positive for the abnormal gene. Every wrong food choice, as well as a failure to deal with stress, can speed up the onset of symptoms. This goes for the other direction too. Every good food choice and the ability to manage stress can go a long way to prolonging positive gene expression. Maintaining an appropriate circadian rhythm is critical to overall health for the entire population. Disruption of circadian rhythm has been linked to heart disease, diabetes, obesity, thrombosis and inflammation.”
from “Nature or nurture: Let food be your epigenetic medicine in chronic inflammatory disorders.” by Katarzyna Szarc vel Szica, Matladi N. Ndlovua, Guy Haegemana & Wim Vanden Berghea. Biochem Pharmacol. 2010 Dec 15;80(12):1816-32. doi: 10.1016/j.bcp.2010.07.029. Epub 2010
“Numerous clinical, physiopathological and epidemiological studies have underlined the detrimental or beneficial role of nutritional factors in complex inflammation related disorders such as allergy, asthma, obesity, type 2 diabetes, cardiovascular disease, rheumatoid arthritis and cancer.
Today, nutritional research has shifted from alleviating nutrient deficiencies to chronic disease prevention. It is known that lifestyle, environmental conditions and nutritional compounds influence gene expression. Gene expression states are set by transcriptional activators and repressors and are often locked in by cell-heritable chromatin states. Only recently, it has been observed that the environmental conditions and daily diet can affect transgenerational gene expression via “reversible” heritable epigenetic mechanisms. Epigenetic changes in DNA methylation patterns at CpG sites (epimutations) or corrupt chromatin states of key inflammatory genes and noncoding RNAs, recently emerged as major governing factors in cancer, chronic inflammatory and metabolic disorders. Reciprocally, inflammation, metabolic stress and diet composition can also change activities of the epigenetic machinery and indirectly or directly change chromatin marks. This has recently launched re-exploration of anti-inflammatory bioactive food components for characterization of their effects on epigenome modifying enzymatic activities (acetylation, methylation, phosphorylation, ribosylation, oxidation, ubiquitination, sumoylation). This may allow to improve healthy aging by reversing disease prone epimutations involved in chronic inflammatory and metabolic disorders.”
 from The Toronto Globe and Mail “Eat your broccoli and ward off cancer.” by Leslie Beck, April 05, 2011
from The Toronto Globe and Mail “Eat your broccoli and ward off cancer.” by Leslie Beck, April 05, 2011
“Looks like Mom was right. Broccoli and cauliflower are good for you. According to a recent review, they’re key ingredients in an eating plan – called the epigenetics diet – designed to fend off cancer.
The epigenetics diet also includes soybeans, red grapes and green tea, foods with active ingredients that influence genes involved in the cancer process.
While genetics is the study of inherited genes, epigenetics looks at changes in the activity of genes. Epigenetics investigates how environmental agents – including the foods you eat – influence which genes are turned on or off.
The traditional view of cancer is that the disease is caused by damage to genes and DNA mutations.
But scientists are learning that other forces – diet, stresses, toxins – have the power to change gene activity in healthy, undamaged cells and ultimately alter cancer risk.
Turns out your diet has the potential to reverse negative changes to gene expression, changes that could, over time, lead to cancer as well as other diseases.
The review by scientists from the University of Alabama at Birmingham outlines how specific components in foods can activate genes that suppress tumour growth and silence genes that promote cancer development.
The epigenetics diet, a term coined after the report’s publication in March, is a daily diet that includes food components that turn on or off a gene’s natural defences against cancer. And it’s easy to adopt since the amounts of active ingredients in foods needed for cancer prevention are very achievable.”
“Diet and the epigenetic (re)programming of phenotypic differences in behavior.” by Patrick O. McGowana, Michael J. Meaneya & Moshe Szyf. Brain Research. Volume 1237, 27 October 2008, Pages 12–24.
“Epidemiological data suggest that dietary changes in methyl contents could affect DNA methylation and gene expression programming. Nutritional restriction during gestation could affect epigenetic programming in the brain. These findings provide evidence for a stable yet dynamic epigenome capable of regulating phenotypic plasticity through epigenetic programming.”
from “Falling for This Myth Could Give You Cancer.” by Dr. Mercola, April 11, 2012
“How Nutrition Alters Genetic Expression
A study performed by the Linus Pauling Institute at Oregon State University was showcased at the annual Experimental Biology convention. The study demonstrated how “histone modifications” can impact the expression of many degenerative diseases, ranging from cancer and heart disease to biopolar disorder and even aging itself.
According to Rod Dashwood, a professor of environmental and molecular toxicology and head of LPI’s Cancer Chemoprotection Program “We believe that many diseases that have aberrant gene expression at their root can be linked to how DNA is packaged, and the actions of enzymes such as histone deacetylases, or HDACs. As recently as 10 years ago we knew almost nothing about HDAC dysregulation in cancer or other diseases, but it’s now one of the most promising areas of health-related research.”
In a nutshell, we all have tumor suppressor genes, and these genes are capable of stopping cancer cells in their tracks. These genes are present in every cell in your body, but so are proteins called “histones.” As Dr. Jean-Pierre Issa at the M.D. Anderson Cancer Center explainsv , histones can “hug” DNA so tightly that it becomes “hidden from view for the cell.” If a tumor suppressor gene is hidden, it cannot be utilized, and in this way too much histone will “turn off” these cancer suppressors, and allow cancer cells to proliferate.
Now here’s where epigenetics comes in … certain foods, such as broccoli and other cruciferous vegetables, garlic, and onions contain substances that act as histone inhibitors, which essentially block the histone, allowing your tumor suppressor genes to activate and fight cancer. By regularly consuming these foods, you are naturally supporting your body’s ability to fight tumors.
Certain alternative oncologists also tap directly into the epigenetic mechanism, such as Dr. Nicholas Gonzalez, who uses a three-pronged approach to cancer based primarily on nutrition and detoxification, and Dr. Stanislaw Burzynski, who treats cancer with a gene-targeted approach. His treatment uses non-toxic peptides and amino acids, known as antineoplastons, which act as genetic switches that turn your tumor suppressor genes “on.”
 A Healthy Lifestyle Supports Healthy Genetic Expression
A Healthy Lifestyle Supports Healthy Genetic Expression
So the good news is that you are in control of your genes … You can alter them on a regular basis, depending on the foods you eat, the air you breathe, and the thoughts you think. It’s your environment and lifestyle that dictates your tendency to express disease, and this new realization is set to make major waves in the future of disease prevention — including one day educating people on how to fight disease at the epigenetic level. When a disease occurs, the solution, according to epigenetic therapy, is simply to “remind” your affected cells (change its environmental instructions) of its healthy function, so they can go back to being normal cells instead of diseased cells.
You can begin to do this on your own, long before you manifest a disease. By leading a healthy lifestyle, with high quality nutrition, exercise, limited exposure to toxins, and a positive mental attitude, you encourage your genes to express positive, disease-fighting behaviors.
This is what preventive medicine is all about. It’s not about taking any one particular nutrient as a supplement to fix one specific “part” of your biological machinery… The more people become willing to embrace this simple truth, the healthier everyone will get.”
from Science Daily “Epigenetic Biomarkers May Predict If a Specific Diet and Exercise Regimen Will Work.” May 30, 2013
“Would you be more likely to try a diet and exercise regimen if you knew in advance if it would actually help you lose weight? Thanks to a report published in the June 2013 issue of The FASEB Journal, this could become a reality. In the report, scientists identify five epigenetic biomarkers in adolescents that were associated with a better weight loss at the beginning of a weight loss program. Not only could this could ultimately help predict an individual’s response to weight loss intervention, but it may offer therapeutic targets for enhancing a weight loss program’s effects.
“Successful obesity treatment during adolescence could reduce morbidity at later stages of life and lead to a better quality of life,” said Amelia Marti, Ph.D., Pharm. D., co-author of this study from the Department of Nutrition, Food Science, Physiology and Toxicology at the University of Navarra in Pamplona, Spain. “It is crucial to find new markers for obesity treatment.”
“If you’ve ever wondered why some people seem to do so well on a diet and exercise plan and other fail so miserably, then now we know that the way that genes express themselves (via epigenetics) plays an important role,” said Gerald Weissmann, M.D., Editor-in-Chief of The FASEB Journal. “This report moves us a step closer when we will be able to prescribe a weight loss program tailored to more than just the lifestyle and conditioning level of the patient, but also to his or her particular genetic and epigenetic profile.”
from Charles A. Moss M.D.
“Epigenetics is possibly the most important and powerful new information on how to lose weight and prevent chronic illness that I have seen in my 35 year medical career.”
from The National Institute on Alcohol Abuse and Alcoholism (NIAAA) “Alcohol Metabolism and Epigenetic Changes.” by Samir Zakhari, Ph.D.
“Alcohol can influence gene expression, and specifically epigenetic regulatory processes that modify the activity of genes, through a variety of mechanisms. Some of these are related to the metabolism of alcohol in the cells. In general, several metabolites, such as nicotinamide-adenine dinucleotide (NAD) in its oxidized and reduced forms, acetyl-coenzyme A (acetyl-CoA), and S-adenosylmethionine (SAM) serve as cofactors for numerous reactions in the cell, including reactions related to epigenetic DNA and histone modifications. Changes in metabolite levels may impact epigenetic processes such as DNA methylation and histone acetylation.”
 from “Research shows junk-food diet is passed on to children in genes.” by Grant McArthur, January 15, 2009
from “Research shows junk-food diet is passed on to children in genes.” by Grant McArthur, January 15, 2009
“OVERWEIGHT parents are condemning their children to obesity and disease by damaging their genes with their own poor diets, scientists have found.
World-first research by the Australian team has revealed that obese sweet-tooths are reprogramming the controls over their DNA, passing on fatness and associated disease to generations of the future.
Scientists from the Baker IDI Heart and Diabetes Institute in Melbourne have proved that damage from unhealthy eating is “remembered” in people’s genetic controls, called epigenetics, turning off good genes needed to prevent diabetes, heart disease and other dangerous complications.
Lead researcher Assam El-Osta said the discovery meant eating a chocolate will not only go straight to your hips, but will also sit on your DNA.
“It is this idea that you are what you eat. Perhaps it’s a reflection of what your parents ate, and perhaps what your grandparents ate,” he said.
The scientists proved that a single sugar hit, such as eating a chocolate bar, damaged the controls regulating the body’s genes for two weeks.
But Professor El-Osta warned that regular poor eating meant the damage would last for months or years, and the real problems caused by an unhealthy diet were deferred until later life.
After having proved the impact of high-sugar foods, the Baker IDI team is now focusing efforts to determine if high fat foods, smoking and other lifestyle-related factors also cause long-term damage to genetic controls, which could then be passed along family bloodlines.
“If you had a Big Mac every day or a very poor diet for a month the consequences would be that you immediately put on weight, but long-term complications are that very serious changes would be remembered for many months or even years,” Professor El-Osta said.
The discovery could explain why some people put on weight or become sick after eating junk food more than others.
“This is not all doom and gloom, because we think there is good epigenetic memory as well for individuals who have a good diet, not only for themselves but potentially for future generations,” Professor El-Osta said.”
from “Epigenetics: A New Bridge between Nutrition and Health.” by Sang-Woon Choi and Simonetta Friso. doi: 10.3945/an.110.1004. Adv Nutr vol. 1: 8-16, 2010.
“Nutrients can reverse or change epigenetic phenomena such as DNA methylation and histone modifications, thereby modifying the expression of critical genes associated with physiologic and pathologic processes, including embryonic development, aging, and carcinogenesis. It appears that nutrients and bioactive food components can influence epigenetic phenomena either by directly inhibiting enzymes that catalyze DNA methylation or histone modifications, or by altering the availability of substrates necessary for those enzymatic reactions. In this regard, nutritional epigenetics has been viewed as an attractive tool to prevent pediatric developmental diseases and cancer as well as to delay aging-associated processes. In recent years, epigenetics has become an emerging issue in a broad range of diseases such as type 2 diabetes mellitus, obesity, inflammation, and neurocognitive disorders.”
from Understanding Nutrition “A Primer on Epigenetics and Diet.”
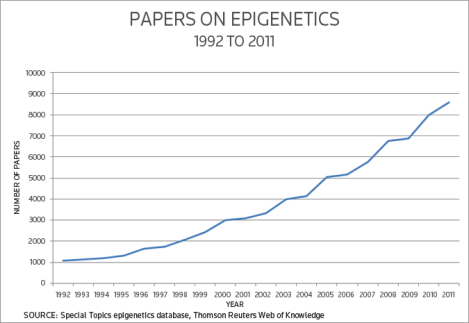
“In the last decade, epigenetics has become one of the hottest topics in science.
The environment largely determines the epigenetic marks present on the DNA. The epigenetic marks improve the body’s ability to adapt to an environment
For example, the body has a set of machinery for processing carbohydrates and different a set of machinery for processing fat. If a person eats mostly carbohydrates, the carbohydrate-processing machinery will be up-regulated and the fat-processing machinery will be down-regulated. If the person switched to a high fat, low carbohydrate diet, they would likely feel sluggish for a few days. They might feel sugar cravings because their body is not yet adapted for using fat as the main fuel source. After a few days, their body would up-regulate the fat-processing machinery and down-regulate the carbohydrate-processing machinery. At that point, they would have adapted to new diet and become efficient at processing fat for fuel.
We are beginning to understand that diet has a big impact on which genes are up – or down-regulated, and this, in turn, has a big impact on our health. A poor diet doesn’t make us unhealthy simply from the consequences of being malnourished or overweight, the diet actually affects the expression of our genes. Certain diets could up-regulate cancer-causing genes and other diets may up-regulate cancer-resistance genes.”
from Epigenetics “Eating for your epigenome.” by Brona McVittie
“If you’re keen to look after your epigenome, then you could try munching on foods that provide the building blocks for methylation in the body. For example, leaf vegetables, peas and beans, sunflower seeds and liver are good sources of folic acid, as are fortified bread and breakfast cereals. Choline comes from eggs, lettuce, peanuts and liver (again! My mother was right after all).
To boost your intake of methionine try spinach, garlic, brazil nuts, kidney beans or tofu. And if you fancy something non-veggie, chicken, beef and fish are all good sources. For zinc, splash out on a plate of oysters. And while you’re on a seafood tip, get some vitamin B12 from fish.”
 from the Biotechnology and Biological Sciences Research Council (BBSRC).”New evidence for epigenetic effects of diet on healthy ageing.” December 2012
from the Biotechnology and Biological Sciences Research Council (BBSRC).”New evidence for epigenetic effects of diet on healthy ageing.” December 2012
“New research in human volunteers has shown that molecular changes to our genes, known as epigenetic marks, are driven mainly by ageing but are also affected by what we eat.
The study showed that whilst age had the biggest effects on these molecular changes, selenium and vitamin D status reduced the accumulation of epigenetic changes, and high blood folate and obesity increased them. These findings support the idea that healthy ageing is affected by what we eat.
Researchers from the Institute of Food Research led by Dr Nigel Belshaw, working with Prof John Mathers and colleagues from Newcastle University, examined the cells lining the gut wall from volunteers attending colonoscopy clinic.
The study volunteers were free from cancer or inflammatory bowel disease and consumed their usual diet without any supplements. The researchers looked for specific epigenetic modifications of the volunteers’ genes that have been associated with the earliest signs of the onset of bowel cancer – an age-related disease.
The investigators studied the relationship between the occurrence of these epigenetic marks at genes known to be affected in cancer, and factors including the volunteers’ age, sex, body size and the levels of some nutrients in the volunteers’ blood. The biggest influence on gene methylation was age. This fits with the fact that the biggest risk factor for bowel cancer is age, with risk increasing exponentially over 50 years old.
The findings, published in the journal Aging Cell, showed that men tended to have a higher frequency of these epigenetic changes than women, which is consistent with men being at a greater risk of bowel cancer. Volunteers with higher vitamin D status tended to show lower levels of methylation, and a similar effect was observed for selenium status. Again, this is consistent with the known links between higher vitamin D and selenium and reduced bowel cancer risk.
The B vitamin folate is essential for health, but in this study, high folate status was associated with increased levels of epigenetic changes linked with bowel cancer. These findings are consistent with some epidemiological studies suggesting that excessive folate intakes may increase risk in some people. The researchers intend to investigate the mechanism for the observed effect of folate on DNA methylation in a follow-up study.
Obesity is also a risk factor for bowel cancer. This study found relationships between body size (height, weight and waist circumference) and epigenetic changes. How excess body weight induces these epigenetic changes, and the consequences for gut health, are currently being investigated at IFR and in Newcastle University.
In summary, the results of this study support the hypothesis that ageing affects the epigenetic status of some genes and that these effects can be modulated by diet and body fatness.”
from Digital Journal “Vegetables in the diet have epigenetic effect to fight cancer.” by Kathleen Blanchard, Mar 8, 2011
Scientists say compounds in vegetables can prevent and even reverse cancer, in what they call an ‘epigenetics’ diet. The researchers say the diet is easily attainable and can prevent aging related diseases and lead to healthier “lives.
Investigators from the University of Alabama at Birmingham explain epigenetics is the study of how gene expression changes over time, leading to cancer and other diseases including Alzheimer’s.
In a study review, published in the journal Clinical Epigenetics, the scientists found compounds in vegetables have been identified from global studies, showing vegetable compounds can help prevent potentially debilitating diseases and boost protection from cancer.
Lead author Syed Meeran, Ph.D., a research assistant professor in Tollefsbol’s UAB Department of Biology laboratory says you don’t have to eat a lot of vegetables to achieve the desired effect. For instance, three cups of green tea might help prevent breast cancer, shown in mouse studies, and a daily cup of broccoli sprouts with the cancer-fighting compound sulforaphane could prevent a variety of cancer types.”
Tollefsbol says, “Our review article has drawn everything together from global studies, and the common theme is that compounds in the epigenetics diet foods can, at the very least, help us lead healthier lives and help our bodies prevent potentially debilitating diseases like breast cancer and Alzheimer’s.”
The notion that vegetables fight cancer and disease is not new. The current study shows why eating vegetables can help prevent cancer and other diseases because compounds in veggies prevent gene aberrations, or epigenetic changes that occur over time in the way genes are expressed.”
 from Examiner.com “Epigenetics Diet – broccoli, cabbage help prevent and treat cancer, Alzheimer’s.” by Amy Rabinovitz, March 12, 2011
from Examiner.com “Epigenetics Diet – broccoli, cabbage help prevent and treat cancer, Alzheimer’s.” by Amy Rabinovitz, March 12, 2011
“An Epigenetics Diet is a daily diet that includes foods that turn on or off a gene’s natural defenses to these cancers and age-related diseases.
Vegetables such as broccoli and cabbage are part of the Epigenetics Diet, as are soybeans, cauliflower, green tea, fava beans, kale, grapes and the spice turmeric.
Meeran says sipping tea compounds called polyphenols in daily amounts that are equivalent to approximately three cups of green tea has been shown to reverse breast cancer in laboratory mice by suppressing the gene that triggers the disease. Similarly, a daily cup of broccoli sprouts, which has sulforaphane as an active compound, has been shown to reduce the risk of developing many cancers.”
from Newsweek Magazine “Controlling Your Genes.” by Sharon Begley, Jan 13, 2010
“Scientists are now making specific, actionable discoveries in epigenetics. This week, for instance, researchers are reporting that eating leafy green vegetables, folate (found in these veggies as well as in some fruits and in dried beans and peas), and multivitamins can affect the epigenetics of genes involved in lung cancer in a way that could reduce the risk of getting the disease, especially from smoking.
As Steven Belinsky of the Lovelace Respiratory Research Institute in Albuquerque and his team describe in a new paper posted online in the journal Cancer Research, they found that eating a lot of leafy green vegetables, folate, and taking multivitamins containing vitamin C, carotenoids, lutein, folic acid, and vitamins A and K was strongly associated with lower methylation of these cancer-related genes.
“A mere 12 servings a month of leafy greens reduced DNA methylation in these genes about 20 percent; taking a multivitamin reduced it almost 50 percent” says Belinsky. “As a result of lower methylation, these beneficial genes—suppressing cancer and repairing DNA—remained on. So it seems that these foods and vitamins can counteract the effect of cigarettes on DNA: although the carcinogens in smoke turn off the beneficial genes, these foods keep them in the game.”
And it isn’t just cancer. A new study from scientists at Cambridge University found a connection between heart disease and DNA methylation. In a nutshell, they found that particular regions of the DNA in the diseased hearts contained DNA-silencing marks—methylation—while healthy hearts did not. “There is already good evidence that these [methylation] marks are strongly influenced by environment and diet,” said Cambridge’s Roger Foo. “We found that this process is different in diseased and normal hearts. Linking all these things together suggests this may be the ‘missing link’ between environmental factors and heart failure.”
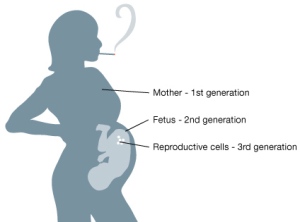
 from The University of Leicester – Virtual Genetics Education Centre “Epigenetics, diet and pregnancy.”
from The University of Leicester – Virtual Genetics Education Centre “Epigenetics, diet and pregnancy.”
“An unbalanced, high-fat, low-protein or energy-restricted diet can modify epigenetic marks, which are associated detrimental health effects in offspring in both animal and human models (Brait et al., 2009, van Straten et al., 2010, Widiker et al., 2010).”